- Home
- »
- Medical Devices
- »
-
U.S. Fluoroscopy Systems Market Size & Share Report, 2033GVR Report cover
![U.S. Fluoroscopy Systems Market Size, Share & Trends Report]()
U.S. Fluoroscopy Systems Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Dedicated Urology Fluoroscopy Tables, Fixed Multipurpose Fluoroscopy for Urology), By Application, By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-874-7
- Number of Report Pages: 80
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Fluoroscopy Systems Market Summary
The U.S. fluoroscopy systems market size was estimated at USD 163.3 million in 2025 and is projected to reach USD 217.5 million by 2033, growing at a CAGR of 4.2% from 2026 to 2033. This robust growth is driven by the increasing demand for real-time and accurate imaging in diagnostic and interventional procedures.
Key Market Trends & Insights
- By product, the fixed multipurpose fluoroscopy for urology segment accounted for the largest revenue share of 62.9% in 2025.
- By application, the endourology & stone management procedures segment held the largest revenue share of 39.6% in 2025.
- By end use, the hospitals segment held the largest revenue share of 48.3% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 163.3 Million
- 2033 Projected Market Size: USD 217.5 Million
- CAGR (2026-2033): 4.2%
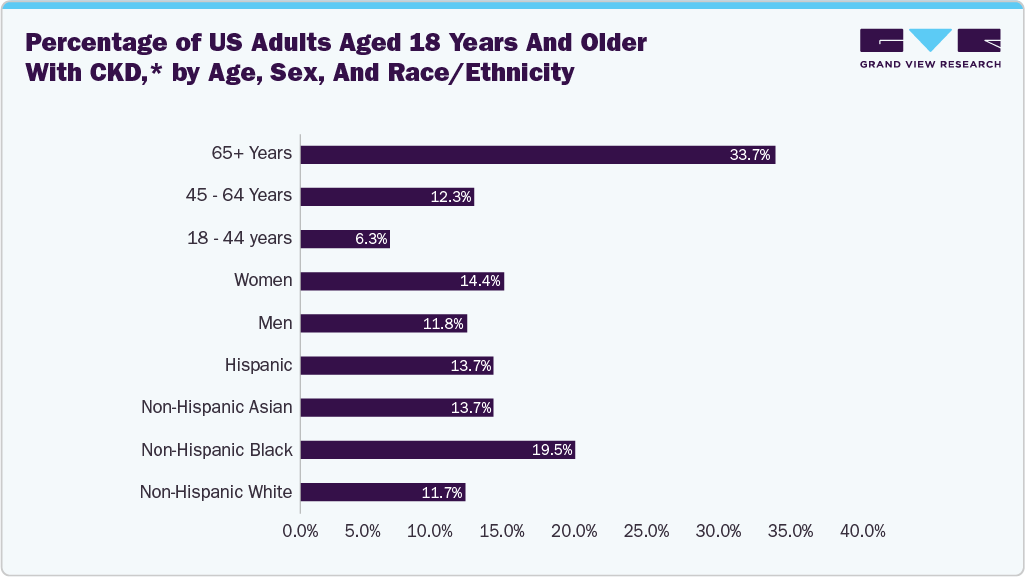
Healthcare providers are focusing more on precision-based treatments, especially in areas such as cardiology, orthopedics, and gastroenterology, where fluoroscopy plays a critical role. The growing preference for minimally invasive procedures is further boosting the adoption of fluoroscopy systems, as these technologies enable better visualization and improved patient outcomes with shorter recovery times. The rising prevalence of kidney and urinary tract diseases plays critical role in the growth of U.S. fluoroscopy systems market. Fluoroscopy provides real-time visualization during minimally invasive procedures, allowing physicians to guide catheters, stents, and other instruments with precision. As chronic kidney disease and urinary disorders continue to affect a significant portion of the U.S. population, healthcare facilities increasingly depend on fluoroscopy systems to support safe and efficient interventions. According to the Centers for Disease Control and Prevention (CDC) report “Chronic Kidney Disease in the United States, 2023,” published in May 2024, more than 1 in 7 U.S. adults (about 35.5 million people) are estimated to have chronic kidney disease, highlighting a significant and growing healthcare burden. The CDC also notes that many individuals remain undiagnosed, increasing the need for early detection and image-guided clinical interventions. This rising disease prevalence supports growing demand for advanced diagnostic and treatment technologies, including fluoroscopy systems used in minimally invasive kidney and urinary tract procedures.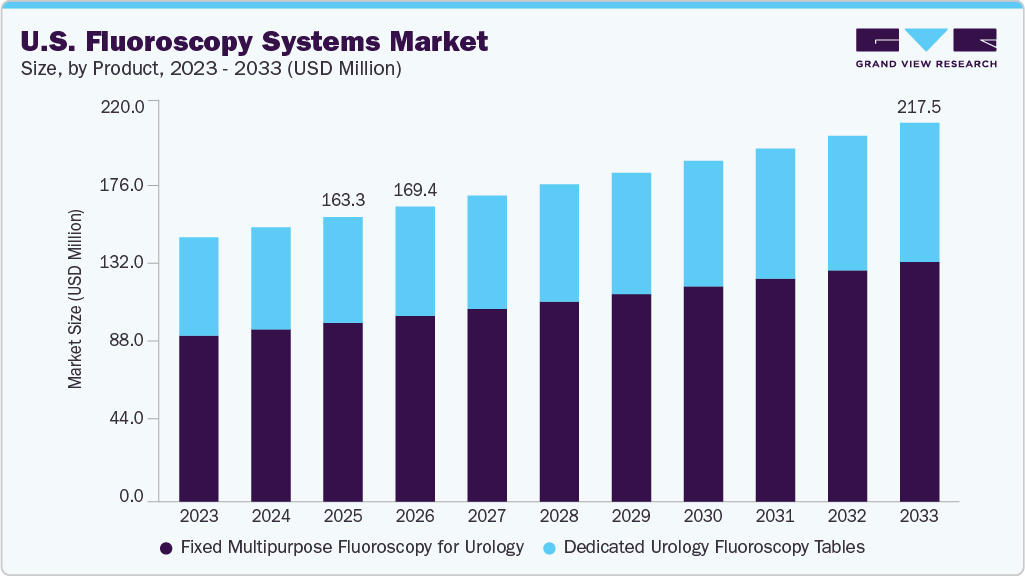
In urology specifically, fluoroscopy is widely used in minimally invasive procedures for kidney stones, urinary obstructions, and related disorders. Kidney stones are a common condition requiring imaging guidance for treatment planning and intervention, and NIDDK notes that about 11% of men and 6% of women in the U.S. experience kidney stones at least once during their lifetime. Fluoroscopy helps urologists visualize the urinary tract in real time during procedures such as ureteroscopy and stent placement, enabling precise instrument navigation while reducing procedural complexity. As the incidence of these conditions rises, the use of fluoroscopy in routine urology practice continues to expand.
Another important factor is that urologic diseases increasingly require efficient, minimally invasive treatment approaches where fluoroscopy plays a central role. Clinical guidance from professional urology organizations emphasizes optimized fluoroscopy use during surgery to improve outcomes while managing radiation exposure, reflecting how essential fluoroscopic guidance has become in modern urologic workflows. Real-time imaging allows physicians to reduce surgical trauma and procedure time, which is particularly valuable for complex kidney and urinary tract interventions. This growing procedural reliance directly supports demand for advanced fluoroscopy systems tailored for urology settings.
Recent population estimates also show a continuing rise in the older age group, reinforcing long-term demand for imaging systems that support efficient and precise treatment workflows. According to a Pew Research Center report published in January 2024, the U.S. centenarian population (people aged 100 and older) is projected to grow from about 101,000 in 2024 to nearly 422,000 by 2054, highlighting a significant long-term demographic shift toward an older population. This trend reflects broader aging dynamics in the U.S., where the number of adults aged 65 and above is also expected to rise substantially over the coming decades. This growing elderly population increases demand for healthcare services, including minimally invasive and image-guided procedures supported by technologies such as fluoroscopy.
Market Concentration & Characteristics
The U.S. fluoroscopy systems market is moderately concentrated, with a few major imaging companies dominating the competitive landscape. Key players such as GE HealthCare, Siemens Healthineers, and Philips Healthcare hold significant market shares due to their strong technological capabilities, broad product portfolios, and established hospital networks. The market is characterized by continuous technological innovation, including digital fluoroscopy, flat-panel detectors, and AI-assisted imaging systems that improve diagnostic accuracy and reduce radiation exposure. High capital investment, strict regulatory approvals, and strong brand presence create entry barriers for new manufacturers. Increasing demand for minimally invasive procedures and image-guided interventions also shapes market competition and product development strategies.
The degree of innovation in the U.S. fluoroscopy systems industry continues to advance steadily. At the RSNA 2022 conference, Philips introduced an expanded AI-enabled imaging portfolio featuring fluoroscopy-related systems such as Fluoroscopy 7000 N and ProxiDiagnost N90. These systems integrate advanced digital radiography with fluoroscopy capabilities to improve clinical decision-making, streamline workflows, and enhance both patient and staff experience as part of Philips’ smart diagnostic imaging solutions.
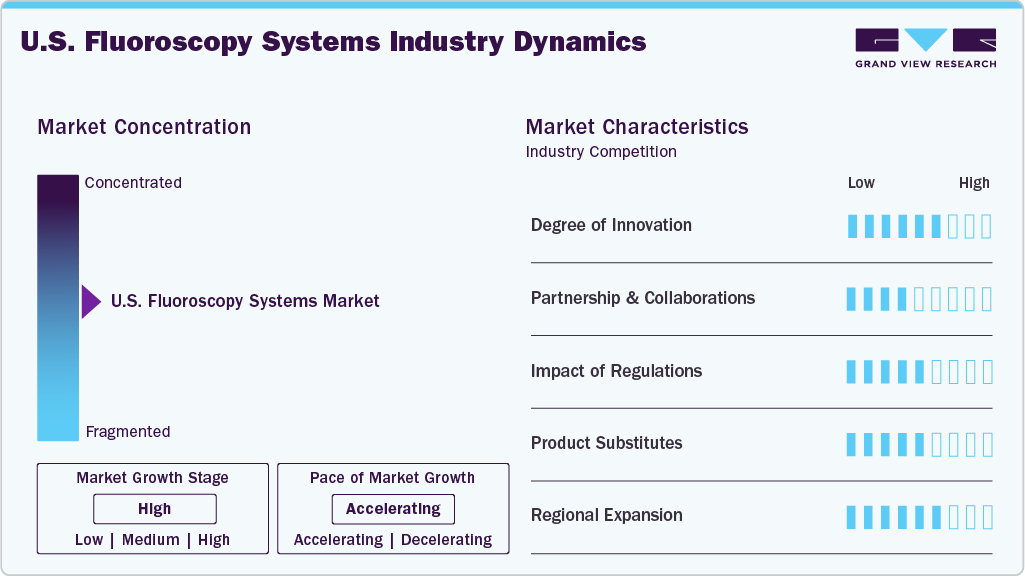
The U.S. fluoroscopy systems industry is experiencing a significant rise in partnerships and collaborations. Companies are increasingly adopting this strategy to expand service offerings and reach a broader customer base. By leveraging combined expertise and capabilities, organizations can deliver improved solutions and services. Additionally, collaborations reduce operational costs and enhance efficiency through shared resources and knowledge. With the growing demand for imaging services, the number of strategic partnerships and collaborations is expected to increase further in over the forecast period.
Stringent regulatory frameworks significantly influence the U.S. fluoroscopy systems industry, ensuring patient safety, device performance, and radiation control. Regulatory authorities such as the U.S. Food and Drug Administration require manufacturers to obtain premarket approval or clearance before commercialization. In addition, standards set by the American College of Radiology and radiation safety guidelines from the National Council on Radiation Protection and Measurements regulate device usage and exposure levels. Compliance with these rules increases development costs and approval timelines but ensures high safety and quality standards. Continuous monitoring, reporting requirements, and periodic inspections further shape product design, manufacturing, and clinical adoption in the fluoroscopy systems market.
Product substitutes in the U.S. fluoroscopy systems industry mainly include alternative imaging technologies that provide comparable diagnostic capabilities with lower radiation exposure or improved imaging efficiency. Modalities such as Computed Tomography (CT), Magnetic Resonance Imaging (MRI), and Ultrasound Imaging are widely used substitutes depending on clinical requirements. CT offers detailed cross-sectional images for complex diagnoses, while MRI provides high-resolution soft-tissue imaging without ionizing radiation. Ultrasound is increasingly preferred for real-time imaging in procedures due to its portability and safety profile. The growing adoption of advanced imaging software and AI-assisted diagnostics further strengthens these alternatives, potentially limiting fluoroscopy demand in certain diagnostic and interventional applications.
In terms of geographic expansion in the U.S. fluoroscopy systems market, UMG / DEL MEDICAL reported in June 2022 that MIS Healthcare installed the UK’s first Apollo DRF remote-controlled fluoroscopy and radiography system at Velindre Hospital in Cardiff. The system introduces advanced flat-panel fluoroscopic imaging with an ergonomic design and an extended focus-detector range. This installation strengthens the hospital’s diagnostic capabilities by supporting both fluoroscopic and radiographic examinations within a single suite, thereby improving clinical workflow efficiency and enhancing overall patient care.
Product Insights
The fixed multipurpose fluoroscopy for urology segment held the largest share of 62.9% in 2025. Adoption of fixed multipurpose fluoroscopy systems for urology is rising across the hospitals and specialty centers in U.S. as healthcare providers are increasingly prioritizing workflow efficiency, procedural precision, and long-term imaging flexibility within a single imaging environment. Fluoroscopy 7000 N — ProxiDiagnost N90, Fluoroscopy 7000 R — CombiDiagnost R90, and Zexira i9 these are some of the examples of fixed multipurpose fluoroscopy systems for urology. Manufacturers also emphasize integrated workflow guidance, simplified controls, and optimized low-dose imaging, aligning with operational pressures to improve efficiency while maintaining patient safety. At the same time, demand for urology procedures is growing due to the rising prevalence of urological conditions in the U.S., with national health data showing that kidney stones affect a significant portion of the population and are increasing over time, thereby expanding the need for imaging-guided urologic interventions.
The dedicated urology fluoroscopy tables segment is expected to grow during the forecast period, due to the increasing number of urological procedures requiring real-time imaging. These specialized systems combine a motorized patient table with fluoroscopic imaging to visualize the urinary tract during procedures such as kidney stone treatment, stent placement, and ureteral interventions. Rising prevalence of kidney stones is a key factor supporting demand. According to the National Institutes of Health, about 11% of men and 6% of women experience kidney stones at least once in their lifetime. Systems such as Uroskop Omnia Max by Siemens Healthineers support efficient workflows through advanced imaging, automated positioning, and dose-reduction technologies.
Application Insights
The endourology & stone management procedures segment dominated the U.S. market with the largest share in 2025. The segment growth is rising mainly because these procedures rely on real-time imaging guidance to safely treat kidney and ureteral stones. During these procedures, fluoroscopy is used to visualize guidewires, catheters, and contrast flow in real time, helping physicians accurately access the urinary tract and manage stones efficiently. Clinical guidance from the American Urological Association highlights minimally invasive endourologic approaches as standard care for many stones, reinforcing the continued need for imaging-guided workflows. In addition, U.S. regulatory standards emphasizing radiation dose display and safer imaging technology are encouraging hospitals to upgrade to newer fluoroscopy systems that support procedural safety and efficiency.
The upper urinary tract imaging and diagnostics segment is expected to grow during the forecast period due to the rising prevalence of kidney stones, urinary tract infections, and other renal disorders that require precise imaging for diagnosis and treatment planning. Increasing adoption of minimally invasive urological procedures has further increased the demand for real-time imaging technologies such as fluoroscopy and advanced radiographic systems. Additionally, the growing geriatric population, which is more susceptible to kidney and urinary tract conditions, is contributing to higher diagnostic volumes. Continuous technological advancements in imaging systems, including improved visualization, lower radiation exposure, and enhanced workflow efficiency, are also supporting the expansion of upper urinary tract imaging in hospitals and specialized urology centers.
End Use Insights
The hospitals segment accounted for the largest share of the U.S. fluoroscopy systems market in 2025 due to its central role in performing a high volume of diagnostic and interventional procedures that require advanced imaging infrastructure. According to the American Hospital Association, the U.S. has nearly 6,100 hospitals with over 35 million annual admissions, creating a substantial demand for imaging equipment procurement and replacement. Hospitals serve as key centers for interventional radiology, orthopedic, urology, and cardiovascular procedures where real-time imaging guidance is essential. In addition to purchasing new systems, hospitals invest in upgrades focused on dose reduction, workflow optimization, and digital integration with hospital IT platforms. Moreover, supportive reimbursement policies such as the CMS Hospital Outpatient Prospective Payment System, which increased outpatient hospital payment rates by 2.6% in CY 2026, continue to encourage hospitals to invest in advanced imaging technologies.
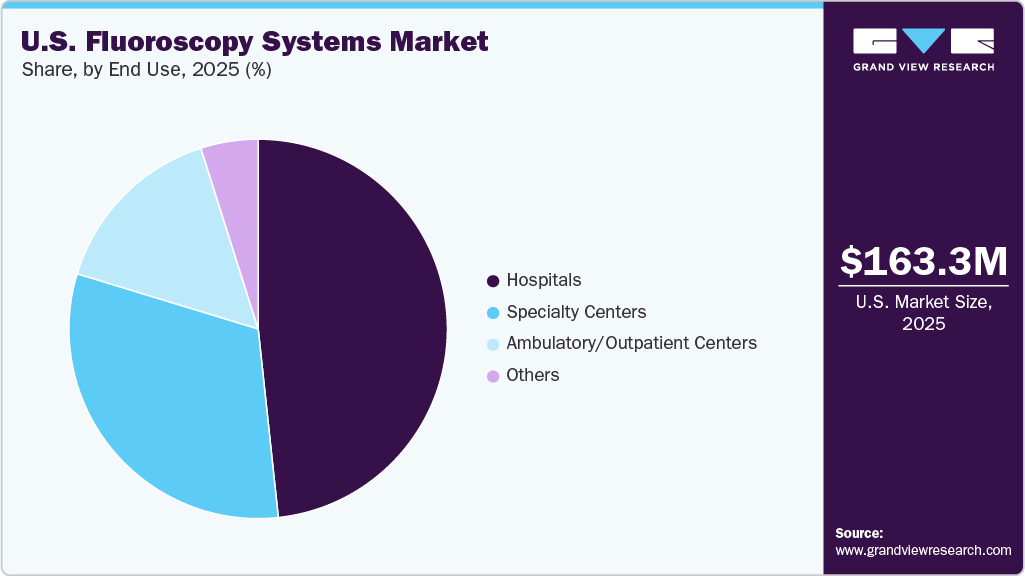
The ambulatory and outpatient centers segment is expected to register the fastest CAGR during the forecast period, due to the growing shift of medical procedures from inpatient hospitals to cost-effective outpatient settings. Ambulatory surgical centers (ASCs) are expanding rapidly, supported by favorable reimbursement policies and increasing procedure volumes. CMS policies influence nearly 6,100 ASCs across the U.S., highlighting a significant and expanding base of facilities that rely on real-time imaging guidance. These centers focus on operational efficiency, shorter patient stays, and reduced treatment costs, increasing demand for compact and user-friendly fluoroscopy systems. According to MedPAC, Medicare-certified ASCs reached around 6,299 in 2023, reflecting steady growth and rising investment in outpatient healthcare infrastructure.
Key U.S. Fluoroscopy Systems Company Insights
The U.S. fluoroscopy systems market is highly competitive, with several established players actively pursuing strategic initiatives to strengthen their market presence. Companies are increasingly focusing on partnerships, collaborations, mergers, and acquisitions to enhance fluoroscopy technologies and improve patient care solutions. For example, in July 2024, Guerbet announced the sale of its urological imaging business to DEL Medical. This transaction transferred key product lines and technologies to DEL Medical, helping expand its footprint in the diagnostic imaging sector, including urology and fluoroscopy applications. By integrating Guerbet’s established urological imaging portfolio and related support services, DEL Medical aims to strengthen its product offerings. This development reflects ongoing consolidation within the imaging industry and highlights efforts to expand capabilities in fluoroscopic and diagnostic imaging systems.
Key U.S. Fluoroscopy Systems Companies:
- GE HealthCare
- Siemens Healthcare GmbH
- Koninklijke Philips N.V.
- Canon Medical Systems
- Shimadzu Corporation
- Dornier MedTech
- UMG / DEL MEDICAL
Recent Developments
-
In August 2025, GE HealthCare Paris Regional Health announced that it has added the advanced GE P180 Fluoroscopy System to its diagnostic imaging services, enhancing its ability to perform a wide range of procedures with superior imaging quality, lower radiation exposure, and improved workflow, marking a significant upgrade in patient care and diagnostic efficiency
-
In January 2024, Shimadzu Medical Systems USA announced that its FLUOROspeed X1 radiographic/fluoroscopy system has received HCAI Special Seismic Certification pre-approval, validating its earthquake-resistant design and enabling safer deployment in seismically active healthcare facilities. The certification supports the system’s use in environments where enhanced structural integrity is required for consistent clinical performance.
-
In July 2023, Canon Medical Systems announced the launch of its Zexira i9 digital X-ray RF system, a versatile multi-purpose fluoroscopy-capable imaging platform featuring a flat panel detector and advanced image processing to deliver high-quality images at low radiation dose and enhanced clinical workflow. The system allows field-of-view adjustment during procedures without moving the table or X-ray tube and includes optional tomosynthesis, DSA, and extended imaging functions for a broad range of fluoroscopic examinations.
U.S. Fluoroscopy Systems Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 169.4 million
Revenue forecast in 2033
USD 217.5 million
Growth rate
CAGR of 4.2% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Product, application, and end use
Country scope
U.S.
Key company profiled
GE HealthCare; Siemens Healthcare GmbH; Koninklijke Philips N.V.; Canon Medical Systems; Shimadzu Corporation; Dornier MedTech; UMG / DEL MEDICAL
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Fluoroscopy Systems Market Report Segmentation
This report forecasts revenue growth at country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. fluoroscopy systems market report based on product, application, and end use.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Dedicated urology fluoroscopy tables
-
Fixed multipurpose fluoroscopy for Urology
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Upper Urinary Tract Imaging & Diagnostics
-
Endourology & Stone Management Procedures
-
Lower Urinary Tract Studies
-
Interventional Urology
-
Post-operative/Follow-up Fluoroscopic Assessments
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Not-for-Profit
-
For Profit
-
-
Specialty Centers
-
Ambulatory/Outpatient Centers
-
Others
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.