- Home
- »
- Medical Devices
- »
-
U.S. Biological Indicators Market Size, Industry Report, 2033GVR Report cover
![U.S. Biological Indicators Market Size, Share & Trends Report]()
U.S. Biological Indicators Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Self-Contained, Strips, Cards / Rapid Readout BIs), By Method (Steam Sterilization, Ethylene Oxide Sterilization), By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-874-5
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Biological Indicators Market Summary
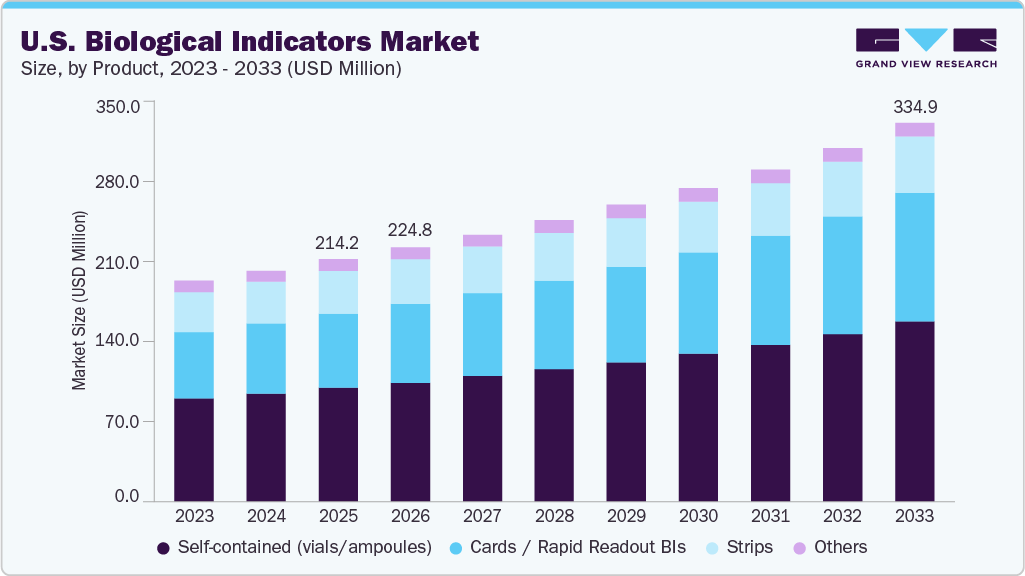
The U.S. biological indicators market size was estimated at USD 214.15 million in 2025 and is projected to reach USD 334.96 million by 2033, growing at a CAGR of 5.87% from 2026 to 2033. The market is primarily driven by the growing emphasis on sterilization validation and infection control across the healthcare and life sciences industries.
Key Market Trends & Insights
- By product, the self-contained segment accounted for the largest market revenue share in 2025.
- By method, the steam sterilization segment accounted for the largest market revenue share in 2025.
- By end use, the hospitals & clinics segment accounted for the largest market revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 214.15 Million
- 2033 Projected Market Size: USD 334.96 Million
- CAGR (2026-2033): 5.87%
Increasing volumes of surgical procedures and hospital-acquired infection (HAI) prevention initiatives are pushing hospitals, pharmaceutical manufacturers, and medical device companies to adopt reliable sterilization monitoring solutions. Strict regulatory requirements from organizations such as the U.S. Food and Drug Administration and the Centers for Disease Control and Prevention mandate routine monitoring of sterilization processes using biological indicators to ensure their effectiveness in hospitals, laboratories, and pharmaceutical manufacturing facilities.The U.S. biological indicators industry is driven by rising surgical procedure volumes and the growing emphasis on effective sterilization monitoring across healthcare facilities. As surgical procedures continue to grow in hospitals, ambulatory surgical centers, and specialty clinics, the need to ensure proper sterilization of surgical instruments becomes critical to prevent healthcare-associated infections (HAIs) and maintain patient safety. Biological indicators play a key role in validating sterilization processes such as steam, ethylene oxide, and hydrogen peroxide, ensuring that sterilization cycles effectively eliminate microbial contaminants.
In addition, the growth in elective cosmetic surgeries is further increasing the frequency of sterilization cycles. According to the 2024 Plastic Surgery Statistics Report published by the American Society of Plastic Surgeons, breast augmentation procedures increased from 304,181 in 2023 to 306,196 in 2024, reflecting a 1% year-on-year rise. This increasing surgical throughput is expected to drive the routine adoption of biological indicators as part of standard sterilization monitoring practices in the U.S. healthcare system.
The growth of the U.S. biological indicators industry is supported by rising investments in pharmaceutical and medical device manufacturing nationwide. Several companies are expanding their production capabilities to meet rising domestic demand and comply with stringent regulatory standards for sterile manufacturing. For instance, in April 2025, BDR Pharmaceuticals announced plans to expand its U.S. operations through the acquisition of manufacturing facilities, supported by an investment of USD 100 million.
Similarly, in June 2025, UCB revealed plans to establish a new biologics manufacturing facility in the U.S., with an estimated economic impact of approximately USD 5 billion. Such investments are increasing the need for validated sterilization processes within pharmaceutical and medical device production. Biological indicators play a critical role in confirming the effectiveness of sterilization for products such as injectable drugs, vials, and syringes in pharmaceutical manufacturing, as well as for implantable medical devices, including pacemakers, stents, and orthopedic implants. As a result, the expansion of sterile manufacturing infrastructure in the U.S. is expected to drive consistent demand for biological indicators used in sterilization monitoring.
The increasing availability of advanced automated sterilization monitoring technologies is expected to support the growth of the U.S. biological indicators industry. Healthcare facilities, particularly sterile processing departments (SPDs), are under growing pressure to improve operational efficiency while maintaining strict compliance with regulatory sterilization standards. As a result, many hospitals and healthcare systems are adopting automated solutions that reduce manual, time-consuming monitoring procedures and enhance workflow efficiency. Industry players are introducing innovative systems that enable faster and more reliable sterilization verification.
For instance, the STERRAD VELOCITY BI/PCD System developed by Advanced Sterilization Products offers one of the fastest biological indicator readout times of approximately 15 minutes. The system integrates automated features that streamline several stages of the monitoring process, including guided biological indicator processing, automatic generation of audit-ready documentation, storage of up to 22,000 BI results, and correlation of sterilizer loads with monitoring data. Such rapid and automated processing can reduce instrument quarantine times by up to 38%, enabling faster turnaround of sterilized instruments and supporting improved sterilization workflow management in U.S. healthcare facilities.
Market Characteristics & Concentration
The degree of innovation in the U.S. biological indicators industry is moderate to high, driven by the need for faster sterilization validation, improved workflow efficiency, and strict regulatory compliance in healthcare and pharmaceutical manufacturing facilities. Market participants are increasingly focusing on developing rapid-readout biological indicators, automated monitoring systems, and digital data management solutions that reduce verification time for sterilization cycles while improving accuracy and traceability. Innovations such as enzyme-based rapid indicators and integrated incubation and reader systems enable healthcare facilities to obtain sterilization results within minutes rather than hours, significantly improving instrument turnaround in sterile processing departments.
The level of merger and acquisition (M&A) activity in the U.S. biological indicators industry is moderate to high, as companies increasingly pursue acquisitions and strategic partnerships to strengthen their sterilization monitoring portfolios, expand technological capabilities, and enhance their presence in infection prevention solutions. Major industry participants such as STERIS, Mesa Laboratories, 3M, and Getinge have engaged in acquisitions to broaden their sterilization monitoring product offerings and integrate advanced biological indicator technologies. For instance, Mesa Laboratories acquired the sterilization indicators business of GKE-GmbH in 2023 to strengthen its product portfolio and global distribution capabilities.
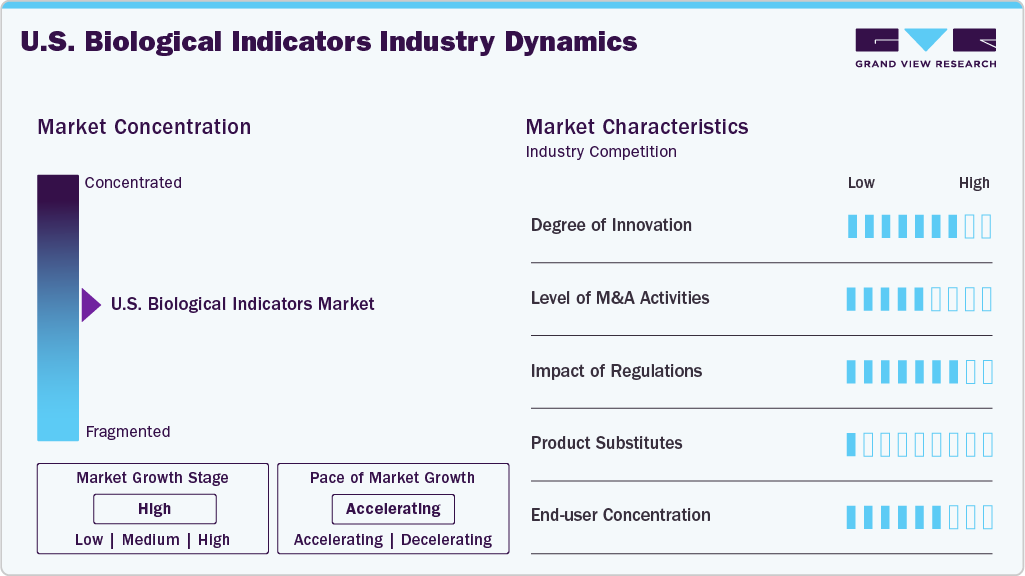
The impact of regulation in the U.S. biological indicators industry is significant, as strict sterilization validation and infection control standards drive consistent demand for reliable biological indicator products. Regulatory authorities such as the U.S. Food and Drug Administration (FDA) and standard-setting bodies such as the Centers for Disease Control and Prevention (CDC) and the Association for the Advancement of Medical Instrumentation (AAMI) establish comprehensive guidelines for sterilization monitoring in healthcare facilities, pharmaceutical manufacturing, and medical device production. These regulations require routine sterilization validation using biological indicators to confirm the effectiveness of sterilization processes such as steam, ethylene oxide, and hydrogen peroxide. Compliance with standards such as International Organization for Standardization (ISO) sterilization protocols further reinforces the mandatory use of biological indicators during sterilization cycles.
The availability of product substitutes in the U.S. biological indicators industry is moderate, as alternative sterilization monitoring methods, such as chemical indicators and mechanical indicators, are widely used alongside biological indicators. Chemical indicators provide rapid visual confirmation that sterilization parameters such as temperature or exposure conditions have been reached, while mechanical indicators monitor physical sterilizer parameters such as pressure, time, and temperature. However, these substitutes cannot directly confirm microbial inactivation, which is the primary function of biological indicators.
Product Insights
The self-contained segment accounted for the largest market revenue share in 2025. This growth is largely attributed to the product’s convenience, ease of use, and lower risk of contamination, as the spore carrier and growth medium are integrated into a single unit. Such a design simplifies sterilization validation processes for hospitals, pharmaceutical manufacturers, and medical device companies across the U.S., improving operational efficiency and reliability. In addition, the growing demand for rapid, user-friendly sterilization monitoring solutions in sterile processing departments and manufacturing facilities is driving the adoption of self-contained biological indicators.
The segment’s growth is further supported by stringent regulatory requirements from agencies such as the U.S. Food and Drug Administration and guidelines from the Centers for Disease Control and Prevention that emphasize validated sterilization monitoring practices. Moreover, ongoing advancements in rapid-readout technologies and the need for faster instrument turnaround in healthcare facilities are expected to further accelerate the adoption of self-contained biological indicators across the U.S. market.
Method Insights
The steam sterilization segment accounted for the largest market revenue share in 2025. This dominance can be attributed to its proven effectiveness, safety, and cost-efficiency in eliminating highly resistant microorganisms. Steam sterilization remains the most widely adopted sterilization method across U.S. healthcare facilities, particularly for surgical instruments in hospitals and ambulatory surgical centers, as well as for reusable laboratory glassware and equipment. Steam autoclaves are commonly used in operating rooms and central sterile supply departments (CSSDs) to ensure proper sterilization and maintain patient safety standards.
In addition, the pharmaceutical and medical device manufacturing industries in the U.S. rely heavily on steam sterilization to validate aseptic processing and sterilize production equipment and culture media. The widespread acceptance of this method, supported by regulatory guidance from organizations such as the U.S. Food and Drug Administration and the Centers for Disease Control and Prevention, has further reinforced the leading position of steam sterilization in the U.S. biological indicators industry.
The hydrogen peroxide sterilization segment is expected to witness at the fastest CAGR over the forecast period. This growth is primarily driven by increasing regulatory and institutional emphasis on sterilization methods that effectively eliminate resistant pathogens while remaining compatible with heat- and moisture-sensitive medical devices. Hydrogen peroxide sterilization offers a low-temperature, residue-free solution, making it suitable for complex instruments commonly used in U.S. healthcare facilities.
Furthermore, the rising concern over healthcare-associated infections (HAIs) and the need for stricter sterilization validation practices across hospitals, laboratories, and pharmaceutical manufacturing facilities are accelerating adoption. Continuous technological advancements in vaporized and plasma hydrogen peroxide sterilization systems, offered by companies such as Advanced Sterilization Products and STERIS plc, are also improving cycle efficiency, reducing turnaround times, and enabling better integration into sterile processing workflows, thereby supporting the rapid growth of this segment in the U.S.
End Use Insights
The hospitals & clinics segment led the market with the largest revenue share of 38.26% in 2025. This dominance is primarily driven by the high volume of surgical procedures and the ongoing need to sterilize critical medical instruments, including surgical tools, endoscopes, and implantable devices. Hospitals serve as the main centers for advanced medical treatment, requiring strict compliance with sterilization standards and infection-prevention protocols. As a result, biological indicators are widely used to validate sterilization processes and ensure patient safety. Furthermore, the growing incidence of healthcare-associated infections (HAIs) and increasing regulatory oversight on sterilization practices across U.S. healthcare facilities have further strengthened the adoption of biological indicators in hospitals and clinics, reinforcing their leading position in the market.
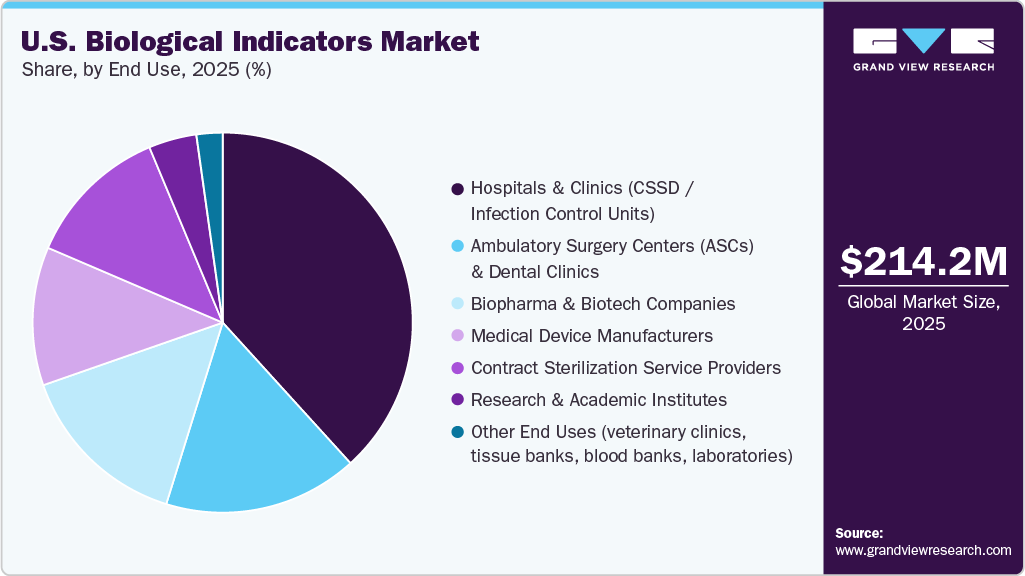
The ambulatory surgical centers (ASCs) & dental clinics segment is expected to witness at the fastest CAGR over the forecast period. This growth is driven by the growing shift toward outpatient care, which offers greater cost efficiency, convenience, and faster recovery than traditional hospital settings. ASCs and dental clinics are performing an increasing number of minimally invasive and routine procedures that require the strict sterilization of reusable instruments. As a result, these facilities are increasingly adopting rapid biological indicators and advanced sterilization monitoring systems to maintain effective infection control while ensuring efficient instrument turnaround. In addition, supportive reimbursement policies, the rising prevalence of chronic and dental conditions, and ongoing investments to expand outpatient care infrastructure across the U.S. are further accelerating the growth of this segment.
Key U.S. Biological Indicators Company Insights
The U.S. biological indicators industry is moderately consolidated, with a few established dental biomaterial manufacturers holding a significant share due to strong brand recognition and wide distribution networks. In addition, several smaller and specialized companies compete by offering niche or innovative graft materials. Market share is largely influenced by product performance, clinician preference, pricing strategies, and long-term relationships with dental clinics and implant specialists.
Key U.S. Biological Indicators Companies:
- Solventum (3M Health Care)
- STERIS
- Mesa Laboratories
- Getinge
- ASP (Fortive)
- Propper Manufacturing Co., Inc.
- Terragene S.A.
- Hu-Friedy
- Medline Industries, LPC
- Ecolab Inc.
- H.W. Andersen Products Ltd.
- True Indicating
Recent Developments
-
In June 2025, Solventum introduced its Attest Super Rapid Vaporized Hydrogen Peroxide (VH₂O₂) Clear Challenge Pack. This ready-to-use test combines two previously FDA-cleared indicators, a biological indicator (BI) for verifying microbial inactivation and a chemical indicator (CI) for confirming proper sterilizer function, into a single-use pack with a transparent container for easy and reliable sterilization validation.
-
In April 2024, 3M finalized the spin-off of its healthcare division, officially launching Solventum Corporation as an independent entity. The company is now listed on the New York Stock Exchange under the ticker symbol SOLV.
-
In October 2023, ASP announced a significant expansion of its Biological Indicators portfolio with new Steam Monitoring products. These innovations are designed to help Sterile Processing Departments (SPDs) enhance sterilization assurance with greater efficiency and confidence in results. ASP’s expanded SM portfolio aims to improve workflow through integrated solutions within Sterile Processing Departments and Central Sterilization Supply Departments (CSSDs).
U.S. Biological Indicators Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 224.75 million
Revenue forecast in 2033
USD 334.96 million
Growth rate
CAGR of 5.87% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Segments covered
Product, method, end use
Report coverage
Revenue, competitive landscape, growth factors, and trends
Regional scope
U.S.
Key companies profiled
Solventum (3M Health Care); STERIS; Mesa Laboratories; Getinge; ASP (Fortive); Propper Manufacturing Co., Inc.; Terragene S.A.; Hu-Friedy; Medline Industries, LP ; Ecolab Inc.; H.W. Andersen Products Ltd.; True Indicating
Customization scope
Free report customization (equivalent to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Biological Indicators Market Report Segmentation
This report forecasts revenue growth at the country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. biological indicators market report based on the product, method, and end use:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Self-Contained (Vials/Ampoules)
-
Strips
-
Cards / Rapid Readout BIs
-
Others
-
-
Method Outlook (Revenue, USD Million, 2021 - 2033)
-
Steam Sterilization
-
Ethylene Oxide Sterilization
-
Hydrogen Peroxide Sterilization
-
Irradiation Sterilization (Gamma, E-beam)
-
Dry Heat Sterilization
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals & Clinics (CSSD / Infection Control Units)
-
Ambulatory Surgery Centers (ASCs) & Dental Clinics
-
Biopharma & Biotech Companies
-
Medical Device Manufacturers
-
Contract Sterilization Service Providers
-
Research & Academic Institutes
-
Other End Uses
-
Frequently Asked Questions About This Report
b. The U.S. biological indicators market size was estimated at USD 214.15 million in 2025 and is expected to reach USD 224.75 million in 2026.
b. The U.S. biological indicators market is expected to witness a compound annual growth rate of 5.87% from 2026 to 2033 to reach USD 334.96 million by 2033.
b. Steam sterilization segment dominated the U.S. biological indicators market with a share of 45.79% in 2025. This is attributable to the product’s convenience, ease of use, and lower risk of contamination, as the spore carrier and growth medium are integrated into a single unit.
b. Some key players operating in the U.S. biological indicators market include Solventum (3M Health Care); STERIS; Mesa Laboratories; Getinge; ASP (Fortive); Propper Manufacturing; Terragene; Hu-Friedy; Medline Industries; Ecolab Inc.; H.W. Andersen Products Ltd.; True Indicating
b. Key factors that are driving the market growth include the growing emphasis on sterilization validation and infection control across the healthcare and life sciences industries. Increasing volumes of surgical procedures and hospital-acquired infection (HAI) prevention initiatives are pushing hospitals, pharmaceutical manufacturers, and medical device companies to adopt reliable sterilization monitoring solutions
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










